Survey of Protists
AUSTRALIAN CURRICULUM ALIGNMENT
- Identify, research and construct questions for investigation; propose hypotheses; and predict possible outcomes (ACSBL030)
- Conduct investigations, including microscopy techniques, real or virtual dissections and chemical analysis, safely, competently and methodically for the collection of valid and reliable data (ACSBL032)
BACKGROUND
The term Protist describes a group of unicellular (or multicellular without tissues) eukaryotic organisms. Classified by their mode of locomotion, Protist also describes protozoa and other eukaryotes with plant-like characteristics. These microscopic organisms produce through cell division and are common on all continents. Protists have previously been classified under Domain Eukarya, Kingdom Protista, and divided into phyla based primarily on their mode of locomotion for some time. However, Protista was not a very accurate grouping as mode of locomotion is somewhat irrelevant to these evolutionary relationships. Currently, the term Protist describes a group of unicellular (or multicellular without tissues) eukaryotic organisms. Protists include organisms with some animal-like characteristics, such as; Protozoa and those with plant-like characteristics, such as; algae. Protists are organised across many different clades and are not necessarily closely related.
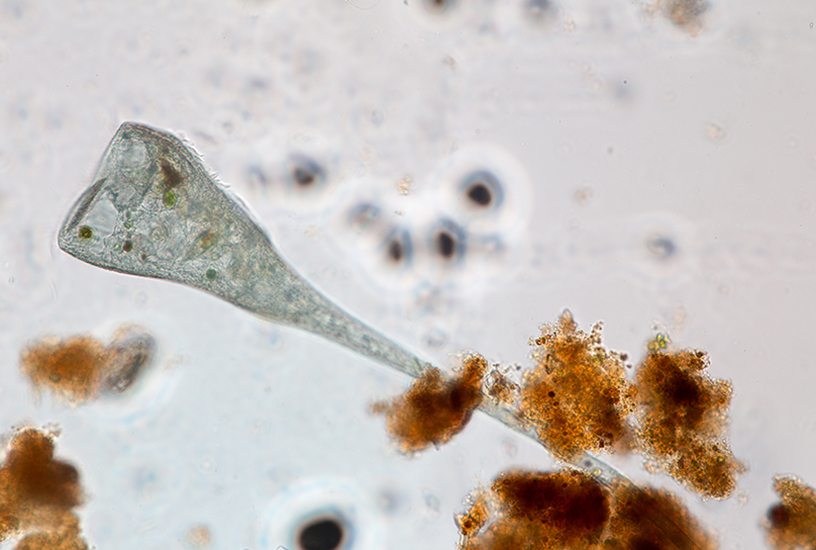
This practical provides an excellent opportunity for students to observe microscopic organisms and compare a variety of unique traits among them. It is perfect for exploring the connection between environment and evolutionary traits as well as how form relates to function. This practical provides an introduction into Protists and basic lab observation practices. Students will observe a series of Protists under a microscope and record their observation data. Students will be required to identify whether the sample is unicellular or colonial, the shape, the structures/ movements, the colour and any specialised features.
PREPARATION - BY LAB TECHNICIAN
General Culture Preparations- Loosen the lid on the container as soon as your Protist cultures arrive.
- Aerate the culture by inserting a pipette in the culture, squeezing and releasing the bulb of your pipette up to 5 times.
- Follow the advised storage conditions for each culture.
- To learn more about Protist culture care, visit our Protist Living Organisms Care Guide.
Preparing Workstations
- Provide each workstation with access to the following materials.
- Prepared stained microscope slides of each organism
- Protozoa cultures
- Pipettes
- Microscope slides
- Coverslips
- Easy View slides and holders
METHOD - STUDENT ACTIVITY
- Collect a microscope slide ready to receive the culture.
- Observe the culture within the jar and identify areas where protozoa are concentrated. The protozoa are most likely the bottom of the culture jar.
- To collect a sample from the culture, squeeze the bulb of your dropping pipette and lower the tip into the water. Position the pipette in the area you wish to sample from. Loosen your grip on the bulb to release pressure and draw the sample in.
- Release 2 to 3 drops from the pipette onto the microscope slide. For ciliates, you will need to add a drop of Protoslo to the slide to slow them down.
- If using a glass slide, cover carefully with a coverslip.
- Alternatively, draw up a few mL of culture, and gently squeeze into an easyview slide, cap the end and observe under the microscope
- View the sample using a low-power objective setting on your microscope and attempt to find a Protist for closer observation.
- Once located, centre the Protist in the microscopic field, and rotate the microscope objective to a higher magnification.
- Record any attributes you observe in the organism in a table. Include the shape, structure, colour and specialised features of the organism, as well as whether the Protist is unicellular or colonial.
OBSERVATION AND RESULTS
Below is a table of the Protist samples we stock and their respective attributes. Be aware that students may observe Protist traits differently; however, this data is not necessarily invalid as the table is only a guide of commonly observed characteristics.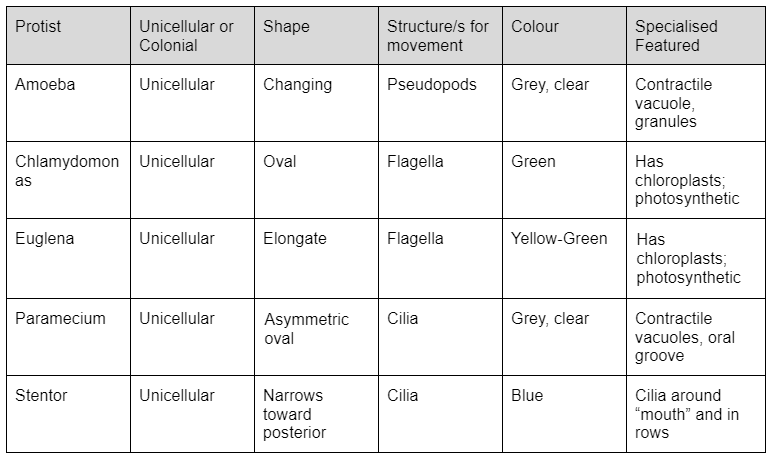
EXTENSION EXERCISE
Easy view slides are a fantastic way of viewing most specimens. The individual culture can be pipetted into the easy view slide and capped ahead of the class, ready for use. It is possible to subculture your living specimens if you want to sustain your cultures beyond the immediate classroom practical. In order to achieve this, simply use the appropriate culture media and follow the instructions. Please remember that the culture media does not contain living specimens.
- Protist culture medium (Paramecium & Amoeba)
- Chlamydomonas culture medium
- Euglena culture medium concentrate
- Stentor culture medium
TEACHER NOTES
- Students may have difficulty collecting the sample. Squirting water into the jar from the pipette or stirring the culture will make collecting a sample more difficult. We recommend preparing the cultures in easy view slides ahead of time, to avoid any mishap, where possible.
- Amoeba can be difficult for students to find. It may be easier to pour the Amoeba culture into a petri dish and let the contents settle. Advise students to gently place the petri dish (on a black background) under a stereo microscope and look through the microscope. Students should then be able to see a ‘fluffy’ creamy-white substance. This is not the Amoeba, but rather their food source. To find the Amoeba hiding underneath, students can gently move the food away using a pipette. After finding the Amoeba, students should observe bright ‘star’ shaped organisms. These are the Amoeba. Whilst looking down the microscope, the Amoeba can be individually drawn up in a pipette and placed on a glass slide for viewing at a higher magnification. If you or your students are unsure of whether if it’s an Amoeba, simply sketch what you are able to see under the microscope. Leave the dish in place for 5 minutes then look gain and sketch what you see. If the shape has changed, it is an Amoeba.
 Time Requirements
Time Requirements
- 45 mins
 Material List
Material List
 Safety Requirements
Safety Requirements
- Wear appropriate personal protective equipment (PPE).
- Dispose of Protists by flushing down the sink with tap water. If the water is not chlorinated, pipette 1 mL of household bleach (sodium hypochlorite solution) or isopropanol (rubbing alcohol) into the culture, and wait 15 minutes before flushing down the sink.
- Wash your hands thoroughly before and after working with any microorganisms.
