Photosynthesis

AUSTRALIAN CURRICULUM ALIGNMENT
- Photosynthesis is a biochemical process that in plant cells occurs in the chloroplast and that uses light energy to synthesise organic compounds; the overall process can be represented as a balanced chemical equation (ACSBL052)
- In eukaryotic cells, specialised organelles facilitate biochemical processes of photosynthesis, cellular respiration, the synthesis of complex molecules (including carbohydrates, proteins, lipids and other biomacromolecules), and the removal of cellular products and wastes (ACSBL049)
- In plants, transport of water and mineral nutrients from the roots occurs via xylem involving root pressure, transpiration and cohesion of water molecules; transport of the products of photosynthesis and some mineral nutrients occurs by translocation in the phloem (ACSBL060)
BACKGROUND
Photosynthesis is the process by which plants transform light energy into chemical energy. Light energy is absorbed and the energy is used to convert carbon dioxide, water and other substances into oxygen, glucose and other nutrients. Photosynthesis can be a hard subject for students to comprehend; however, it can be clearly demonstrated using Elodea and a solution of Bromothymol Blue. Elodea is a commonly used aquatic plant in freshwater aquariums that is capable of growing rapidly when introduced to freshwater ecosystems. As the Elodea absorbs carbon dioxide from the pre-prepared solution, the pH changes cause the colour to change. This change will be observable within 30 minutes to an hour. Bromothymol turns blue when exposed to alkaline (basic) conditions, and yellow-green in acidic conditions; making it an excellent pH indicator. At the beginning of this practical, the Bromothymol will be yellow-green as the carbon dioxide (CO2) is pre-added. Carbon dioxide reacts with water to form carbonic acid in equilibrium, indicating a simultaneous reaction in both directions. As CO2 leaves the system, the system will achieve equilibrium once again as the carbonic acid dissociates. Photosynthesis will remove carbon dioxide from the solution; becoming more basic and turning from yellow to dark green and then to blue.
In this practical, students investigate photosynthesis, the reactants (substances used to create a reaction) and products (products created through the reaction) of the process. This practical provides a visual illustration of photosynthesis that allows students to observe the process in a more tangible way. Students will observe how radiant energy is converted into organic substances that can be stored within the plant to support the growth, reproduction, and metabolism during photosynthesis. Students gain an understanding of the process of photosynthesis, the role of carbon dioxide and how plants metabolise the sugars formed as sunlight, carbon dioxide, and water react.
PREPARATION- BY LAB TECHNICIAN
- Clean the Elodea by rinsing it in pond water or conditioned (aged) tap water to remove any small leaf pieces or sediment.
- Using scissors, cut 2 x 6cm sprigs of Elodea for each group. Keep the Elodea in a bowl filled with pond water for students to easily access.
- Prepare a diluted 0.04% Bromothymol Blue solution.
- Just prior to commencing the lab, pour the Bromothymol Blue into a beaker. Exhale gently into a straw submerged in the solution. This will add carbon dioxide to the solution, changing the pH. Continue to blow bubbles into the liquid until it turns yellow-green. This process should take approximately 15 to 30 minutes.
- Never inhale through the straw or allow the fluid to reach towards your mouth. To avoid this, be careful not to blow too vigorously as it may cause the liquid to splash onto your skin. Wear protective goggles and cover the beaker with a piece of cardboard or aluminium foil.
- Measure and cut 20 cm × 10 cm pieces of aluminium foil pieces to wrap capped tubes.
METHOD - STUDENT ACTIVITY
-
Write your initials on the capped tubes with a marker.
- Write “D” for dark towards the top of one test tube and “L” for light on the other.
- Place a 6-cm sprig of Elodea in each of the tubes.
- Fill both tubes to overflowing with the yellow-green Bromothymol Blue solution using a pipette and replace the cap. Use a piece of paper towel to clean up any spills.
- Observe both tubes as quickly as possible and note what you see.
- Immediately wrap the tube marked “D” with aluminium foil. Cover it carefully so that no light can potentially reach the Elodea.
- Place both tubes in the sunlight or under a light bank.
- After 1 hour, record what you see.
OBSERVATION AND RESULTS
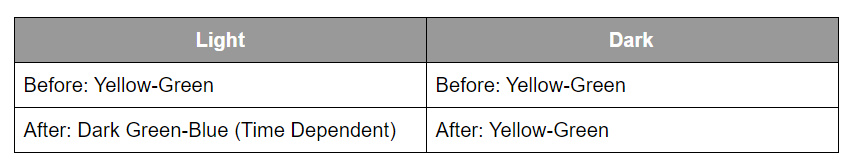
The exposure time to light will alter the colour of the solution. More time will lead to a darker blue. The change in colour is the result of the Elodea photosynthesizing. By photosynthesizing, the Elodea uses the carbon dioxide in the solution. More carbonic acid dissociates to form more carbon dioxide and water. Thus, the solution becomes more basic. The pH indicator, Bromothymol Blue reacts to more the basic solution by turning it green and eventually blue. Below are the expected results for the 2 test tubes.
 Time Requirements
Time Requirements
- 45 mins
 Material List
Material List
-
Bromothymol Blue (200 mL of 0.04%)
- Sprigs of Elodea or Hydrilla (6 cm)
- Aluminum Foil
- Marker or Wax pencil
- 2 Test Tubes with Caps
- Pipette
- Paper Towel
- Light bank (if there is no access to natural sunlight)
- Pond Water (or Aged water)
 Safety Requirements
Safety Requirements
- Wear appropriate personal protective equipment (PPE).
- Know and follow all regulatory guidelines for the disposal of laboratory wastes.
- Wash hands thoroughly before and after handling any chemicals.
- Sterilise work surfaces before and after the practical.
- Bromothymol Blue may stain your hands and clothing; avoid any direct contact.
