Natural Selection in Brine Shrimp

AUSTRALIAN CURRICULUM ALIGNMENT:
-
Natural selection occurs when selection pressures in the environment confer a selective advantage on a specific phenotype to enhance its survival and reproduction; this results in changes in allele frequency in the gene pool of a species (ACSBL090)
BACKGROUND:
Brine shrimp (Artemia Salina) are a species of small crustaceans that are found in saline environments, specifically salt lakes, worldwide. Brine shrimp do not inhabit oceans due to the high presence of predators. Brine Shrimp are able to avoid predators by living in very high saline environments that other aquatic life are not able to. This makes them an excellent model for the study of natural selection and adaptations. When environmental conditions are optimal, female Brine Shrimp produce thin shelled eggs that develop steadily and hatch quickly into live young. Less ideal environmental conditions, such as low oxygen levels or extremely high salinity will trigger females to produce thicker shelled cysts that are covered in a hardened, brown outer layer called a chorion. The chorion maintains the embryos in a dry, oxygen-free environment. These encased embryos can survive for months or even years in this dormant state called diapause. The development of the cysts is suspended until changes in the environmental conditions cause the embryos to develop further, and eventually hatch.
When environmental conditions are ideal once again, the cysts break open and the Brine Shrimp remains attached to the shell, surrounded by a hatching membrane. During the umbrella stage, the nauplius (larval brine shrimp) remains attached to the cyst for a few hours. In the first larval stage, the nauplius feeds on the yolk reserves until it sheds its exoskeleton for the first time. The nauplius begins to feed on algae during the second larval stage. In the 2-3 weeks it takes the Brine Shrimp to reach adulthood it moults approximately 15 times.
Salinity levels within the environment greatly impact population growth of Brine shrimp. In this investigation, you explore how hatching viability is impacted by different saline level environments. To do this, you will attempt to hatch cysts in four different salt concentrations and measure the hatching viability by counting how many nauplius that emerge from the cysts. Brine Shrimp will hatch within 24 - 48 hours making this a great way to quickly observe how Brine Shrimp have adapted to survive in different environmental conditions.
PREPARATION - BY LAB TECHNICIAN
-
To make the 2% salt solution, add 20g of NaCl (salt) to 1L distilled water.
- To make the 1% salt solution, add 10g of NaCl to 1L distilled water.
- To make the 0.5% salt solution add 5g of NaCl to 1L of distilled water.
- Aliquot approximately ¼ of a teaspoon of brine shrimp eggs into vials for each student workstation.
METHOD - STUDENT ACTIVITY
Preparing Cysts for Hatching (Day 1)
-
Using a permanent marker, label 4 petri dishes: 0%, 0.5%, 1.0%, 2.0%.
- Form your hypothesis; for example, ‘If the salinity of the hatching solution is 2%, then more cysts will hatch and thrive.’
- Using the graduated cylinder, measure 30 mL of each saline solution and 30mL of distilled water and pour it into the appropriately labelled Petri dish.
- Collect four microscope slides. Measure and cut four 1.5 cm strips of double-sided tape and gently adhere one of them to each of the microscope slides.
- Lightly touch the fine brush to the side of the dish containing the brine shrimp eggs. Collect 20–30 eggs on the brush. Do not collect too many eggs because you will be required to count them.
- To adhere the eggs to the double-sided tape, lightly press the brush onto the tape on the first microscope slide. Repeat this step for the remaining three microscope slides.
- Using a microscope, count the number of eggs on the first slide. Record this information in the results table.
- Once the eggs have been counted, place this slide into the 0% salt solution Petri dish. Place the slide with the tape side facing up.
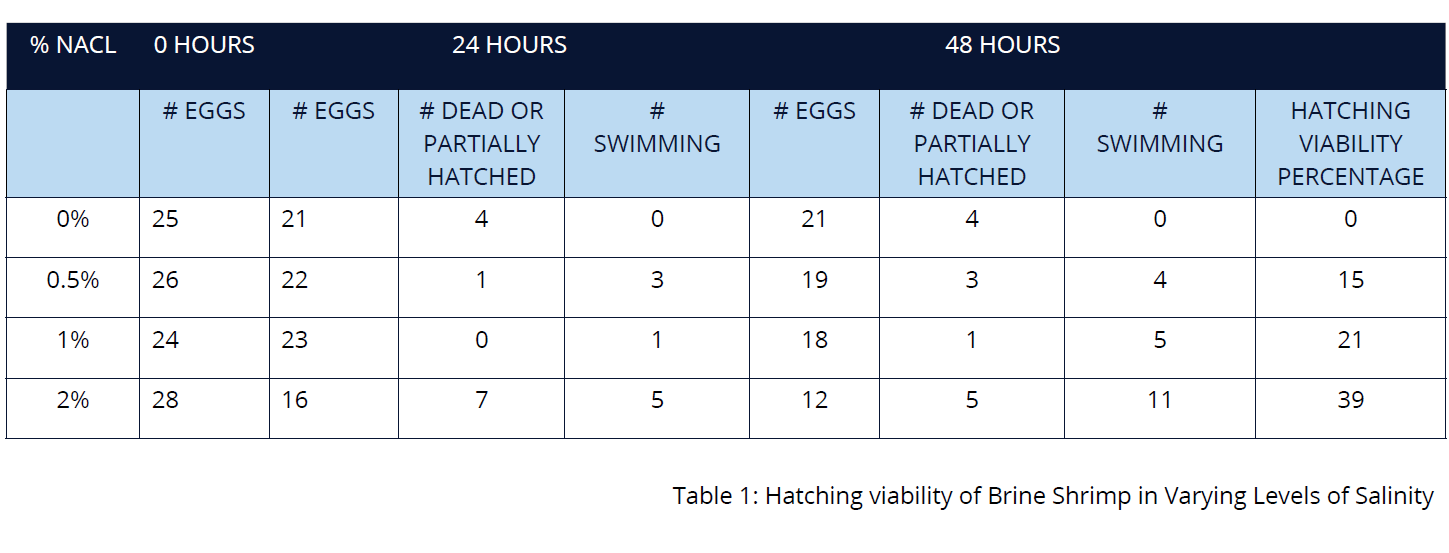
- Count the eggs on each slide and place them in the respective salt solutions. Copy Results Table 1 into your logbook and record the egg count information (at 0 hours).
- Place the Petri dishes under a light bank for 24 hours at room temperature.
Data Collection (Days 2& 3)
-
After 24 hours, examine the contents of each Petri dish under the stereomicroscope. You should see that some brine shrimp have hatched and are swimming in the salt solution. Record the number of eggs, the number of dead or partially hatched eggs and the number of swimming brine shrimp information in your results table.
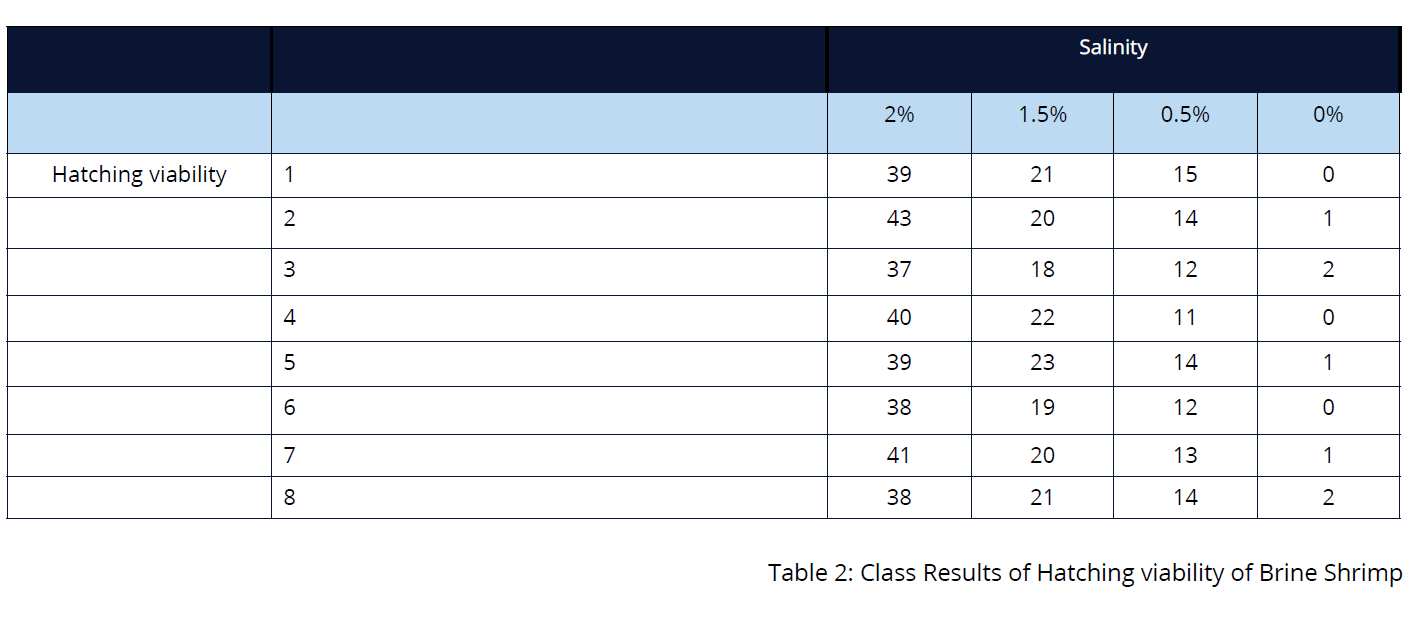
- After 48 hours, examine the contents of the Petri dishes again and record your observations in the results table. Calculate the hatching viability of each dish at 48 hours by dividing the number of shrimp swimming by the initial number of eggs in the Petri dish. Round up your calculations to the nearest hundredth and add this information to the class results table.
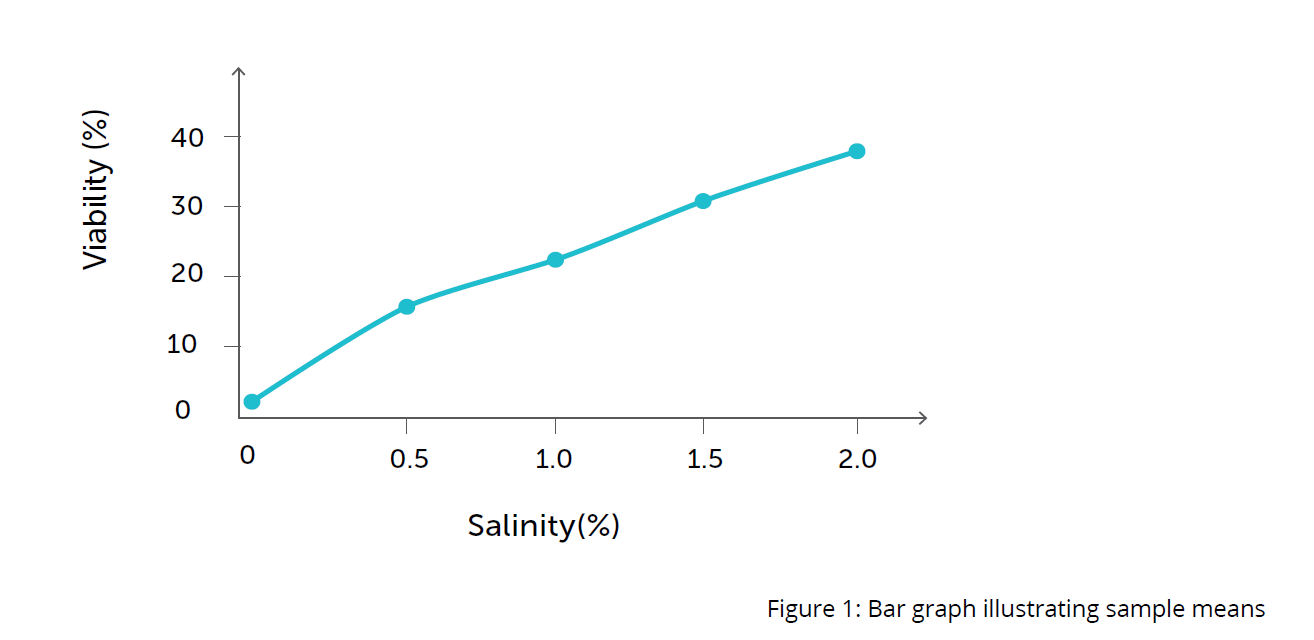
- Draw a bar graph that shows the sample means from the class results.
OBSERVATION AND RESULTS
Below is an example of expected results for your experiment. This is an example only as individual results will vary.
 Mean, standard deviation, and standard error of the mean will vary depending on class data results.
Mean, standard deviation, and standard error of the mean will vary depending on class data results.
INVESTIGATIONS
-
Describe two conditions that were controlled in this experiment.
- The two main conditions controlled in this experiment are temperature and time.
- Which Petri dish had the highest hatching viability? Which had the lowest? Suggest possible reasons for these results.
- The petri dish containing a 2% salinity solution exhibits the highest hatching viability. The petri dish containing a 0% salinity solution has a 0% hatching viability. These results indicate that brine shrimp will not hatch in environments containing no salt and thrive in environments of higher salt concentration.
- Was your hypothesis supported? Explain why.
- Student answers will vary.
- Based on your individual data and the class data, is there enough evidence to conclude that environments of different salinities affect the hatching viability of brine shrimp?
- The individual and class data provides sufficient evidence to conclude that the hatching viability of the shrimp is affected by different salinities. However, a larger data set would provide more conclusive evidence regarding the optimum salinity levels for hatching viability.
EXTENSION EXERCISE
- What other conditions may affect the hatching viability of brine shrimp? Design an experiment to investigate another environmental factor that may impact hatching viability.
 Time Requirements
Time Requirements
- 45 mins
 Material List
Material List
- 4 Petri Dishes
- Salt Water Solutions (0.5%, 1.0 % and 2.0%)
- 1 Fine Brush
- 4 Microscope Slides
- 4 Strips of Double-Sided Tape
- 1 Stereo Microscope (or magnifying glass)
- 1 Permanent Marker
- Distilled Water
- 1 Graduated Cylinder
 Safety Requirements
Safety Requirements
-
Wear appropriate personal protective equipment (PPE).
- Know and follow all regulatory guidelines for the disposal of laboratory wastes.
- Wash hands thoroughly before and after working with any organic materials.
- To dispose of brine shrimp, pour them down the sink. Alternatively, you may like to keep them for further observation.
